Metal Accumulation in Huntington’s Disease
Researchers have observed that many neurodegenerative diseases are associated with the accumulation of heavy metals in the brain. Iron elevation is especially well documented, but increasing evidence associates the accumulation of other metals like copper with neurodegeneration as well. Elevated levels of metal ions may contribute to the generation of free radicals which can lead to neuronal atrophy.
What is the role of iron in the body?^
Iron plays an important role in normal human functioning. It is involved in oxygen transport, electron transport and many metabolic processes. Iron in the body is bound in proteins to prevent excess toxicity. Brain cells like oligodendrocytes, neurons, and microglia all express ferritin, an iron storage protein.
Despite the importance of iron to normal human functioning, excessive accumulation of iron in the brain has many negative consequences. Increased iron can damage cells directly or perturb the cellular environment. Iron accumulation can make cells more vulnerable to toxins, inflammation or apoptosis. Additionally, high iron levels contribute to reactive oxygen species generation and protein aggregation, which lead to damaging oxidative stress.
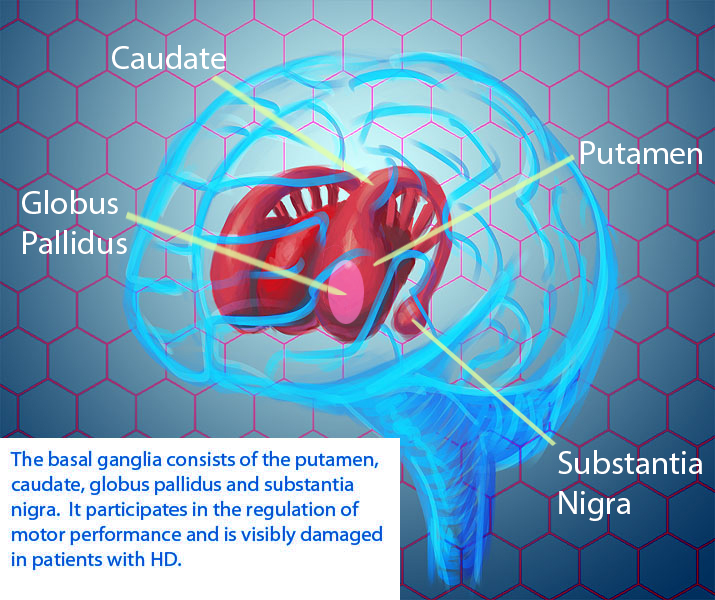
For individuals with Huntington’s disease, increased iron levels have primarily been observed in the basal ganglia. The basal ganglia is a brain region that includes the striatum, internal and external globus pallidus, subthalamic nucleus and substantia nigra. One study using magnetic resonance imaging to quantify iron levels in HD patients found the most noticeable iron accumulation in the striatum and the globus pallidus. For more information about the basal ganglia visit the HOPES Brain Tutorial here.
Iron levels increase early in the disease process and continue to increase with advancing age. The steady accumulation of iron in HD patients suggests that it may play a role in the progression of the disease.
Iron is important for the normal function of neurons and many areas of the brain naturally have high iron content. However, abnormal amounts of iron specifically in the basal ganglia, is a hallmark of neurodegenerative disorders. It has been observed in Parkinson’s disease, Alzheimer’s disease and Friedreich’s ataxia as well as in HD. Although these other neurodegenerative diseases share with HD problems of iron regulation in the basal ganglia, those problems do not manifest identically. For instance, Parkinson’s disease is characterized by iron accumulation in the substantia nigra, which has not been observed in HD.
There are several reasons why iron might accumulate in the course of a neurodegenerative disorder. The iron accumulation might be a direct result of the disease. In the case of HD, scientists do not yet understand why this would occur. But it is also possible that the iron accumulation is a consequence of secondary effects of the disease. For instance, in HD iron accumulation might occur because after neuronal loss (a direct result of HD) the cells that died are replaced by cells with higher iron content (secondary effect). Another alternative is that iron accumulation is facilitated by the breakdown of the blood brain barrier, an event which might allow excess iron from the body to enter the brain.
Do other metals accumulate in HD?^
Researchers have also found that HD is associated with increased copper levels in the brain. Copper levels appear to be higher in the striatum of HD brain tissue. Additionally, studies with transgenic mice found elevated copper levels. In one study, 12 month old R6/2 mice had copper levels increased 26% in the striatum and 51% in the cortex.
Copper has been shown to interact chemically with both the normal and altered huntingtin protein. Copper may contribute to HD pathogenesis by promoting the formation of huntingtin aggregates. To learn more about the role of huntingtin aggregates in HD, click here. Additionally, copper could play a role in HD pathogenesis by changing the conformation of the altered huntingtin protein.
What are the therapeutic implications of metal accumulation?^
The association of accumulation of iron and other metals with HD suggests a couple of treatment strategies.
One strategy is chelation therapy. Chelation therapy is a process for removing heavy metals from the body. It works by introducing a chemical compound into the body that can bind to the unwanted heavy metal. The compound renders the unwanted metal chemically inert by binding to it and thus allows the metal to safely pass through the body without causing any further harm.
Chelation therapy could potentially be used in HD to eliminate some of the excess heavy metals that accumulate in the brain. Chelation therapy is a promising possibility for HD because chelation treatments are widely available and could easily be evaluated for efficacy in HD patients. Research is currently underway to test the efficacy of an iron chelator, M30, and a copper chelator, Coprexa, for HD.
Chelation therapy can have side effects. Most common is a burning sensation at the site where the chelating agent is delivered. Other side effects include headaches, skin irritation, nausea, fatigue, fever and cramps. Rare side effects include drops in blood pressure, kidney and liver problems, low calcium levels, and low blood cell counts. Serious side effects have a low occurrence, but the therapy should always be administered by a trained medical professional.
Another treatment strategy that combats the damaging effects of heavy metals is to target the reactive oxygen species generated by metal accumulation. Reactive oxygen species are one type of free radical. A tremendous amount of research is focused on the potential for antioxidants to combat free radicals. To learn more about free radical damage and the antioxidants that might help fight it, click here.
For Further Reading^
The following readings contain greater detail, but all of them are technical and demand familiarity with scientific language.
- Bartzokis, George et al. “Increased basal ganglia iron levels in Huntington disease.” Archives of Neurology. 56 (1999): 567-564. A study which used magnetic resonance imaging to observe increased iron levels in the basal ganglia of HD patients.
- Dexter, DT et al. “Alterations in the levels of iron, ferritin and other trace metals in Parkinson’s disease and other neurodegenerative diseases affecting the basal ganglia.” 114 (1991): 1953-1975. Authors measured the accumulation of metals in brain tissue from patients with a variety of neurodegenerative diseases..
- Fox, Jonathan H. et al. “Mechanisms of copper ion mediated Huntington’s disease progression.” PLoS One 3 (2007): e334. Presents evidence for copper and iron accumulation in HD.
- Madhavi Thomas and Joseph Jankovic. “Neurodegenerative disease and iron storage in the brain.” Current Opinion in Neurology 17 (2004): 437-442. Compares the differences in iron accumulation across neurodegenerative diseases.
- Zecca, Luigi et al. “Iron, brain ageing and neurodegenerative disorders.” Nature Reviews Neuroscience 5 (2004): 863-873. Comprehensive review of iron’s role in neurodegenerative diseases, also summarizing iron’s importance in the body and the brain.
A. Hepworth, 2/2/09